|
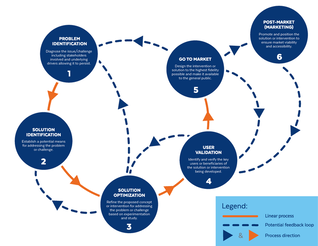
Introduction The drug development process is often presented as a linear process: you achieve one milestone before moving on to the next, with progress taking the developer ever forward. The Food and Drug Administration defines this process as such:
Based on the way that this information is conveyed, you might expect the process to visually represent something like this: Understanding people’s perspectives around male contraception helps guide our work at MCI. Sharing them is a privilege as well as a mandate: it’s of strategic importance to us to facilitate and broadcast the interest for male methods in an effort to present the undeniable market demand for them. Normally this is done through the lens of our grantees, fellows, research community, or even the general public. But lately, we’ve been endeavoring to share the views of MCI’s staff as well. In that spirit, this is a conversation with our Communications & Marketing Director Kevin Shane in which he shares his thoughts about male contraception as well as his interest in working with MCI.
Oleksandr “Sasha” Kirsanov is a PhD candidate in the Geyer laboratory at East Carolina University’s Brody School of Medicine. His work is focused on defining the mechanisms underlying spermatogonial differentiation and meiotic initiation. In this blog post, Sasha shares about his work in the field of contraception, what he hopes to learn as an MCI fellow, and the future outlook of male contraception.
|
Categories
All
Archives
June 2024
|
|
|
Donate to Male Contraceptive InitiativeYour generous donation makes a difference!
|
© Male Contraceptive Initiative. All rights reserved.



 RSS Feed
RSS Feed
